WHS Posters
(WHS-P4) SLI-F06, A FIBROMODULIN-BASED THERAPEUTIC PEPTIDE, ENHANCES WOUND HEALING IN DIABETIC RODENT AND PIG MODELS
Friday, May 17, 2024
7:30 AM - 5:00 PM East Coast USA Time
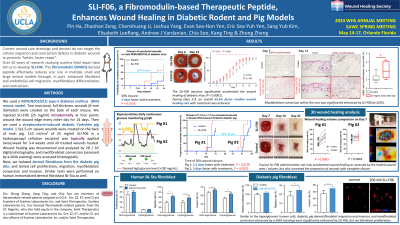
SLI-F06 is a fibromodulin-derived peptide that effectively promoted wound healing and reduced scar size without discernable adverse effects in multiple preclinical and clinical acute wound studies. It functions through, in part, enhancing fibroblast migration, myofibroblast differentiation, and contraction. Since chronic wounds are deficient in cell migration and wound contraction, we sought to test SLI-F06’s ability to heal cutaneous wounds in challenging diabetic models. Because over 90% of adult diabetic patients in the US exhibit type 2 diabetes mellitus (DM) rather than type 1, we used a NONcNZO10/LtJ type 2 DM mouse model for our first set of wound studies. Two excisional, full-thickness wounds (6 mm diameter) were created on the back of each mouse. To simulate human-type repair in the loose-skin mouse, we sewed silicone rings to each wound periphery to “splint” the wound to minimize excessive wound margin contraction. Then, we injected SLI-F06 (25 mg/ml) intradermally at four points around the wound edge (25 ml/point; 100 ml total) every other day for 14 days. We documented wound area healing by digital photography and fitted the data to a quantile mixed-effect model with a 95% confidence interval. These data demonstrated that the SLI-F06 application significantly accelerated the wound healing of diabetic NONcNZO10/LtJ mice (P < 0.0001). Moreover, multiple comparisons applying Šidák correction revealed that SLI-F06 markedly enhanced wound healing from Day 1 to 13 (adj. P < 0.05, N = 12). Particularly, during Days 2-8, the quantile mixed-effect model revealed an overall 65.6% (> 30%) faster median wound healing rate with SLI-F06 treatment. Furthermore, the proportion of wounds with complete closure at day 14 post-injury was increased considerably (Mantel-Cox test P = 0.0156; N = 12) at 83.3% in the SLI-F06 group vs. 41.7% in control. Next, we used a streptozotocin-induced diabetic Yorkshire pig model that simulates certain characteristics of later-stage human type 2 non-insulin dependent DM, such as increased triglycerides and glucose intolerance. We excised 1.5 x 1.5-cm square wounds and topically applied 110 ml/cm2 of 25 mg/ml SLI-F06 in a hydroxypropyl cellulose excipient twice/week. Excitingly, topical SLI-F06 administration not only accelerated wound healing as assessed by the healed wound area, but also increased the proportion of wounds with complete closure (SLI-F06: 15.0 day vs. control: 18.0 day; Mantel-Cox test P = 0.0300, N = 7). These promising preclinical data strongly support the effectiveness of SLI-F06 in diabetic wound management, which can significantly improve the quality of life of diabetic patients suffering from chronic wounds that can lead to amputations and death.

.jpeg)
