WHS Oral Abstracts
(WHS-O.04) SENESCENT-ASSOCIATED EXTRACELLULAR MATRIX PRODUCTION AND DELAYED WOUND HEALING BY SENESCENT DERMAL FIBROBLASTS
Thursday, May 16, 2024
6:00 PM - 6:30 PM East Coast USA Time
Background: After skin injury, a sub-population of the dermal fibroblasts undergo senescence during the early stages of the healing process. While the transient appearance of senescent cells is required for normal wound healing, the accumulation of persistent senescent cells is associated with delayed and chronic wound healing. The mechanisms by which senescent fibroblasts and their associated extracellular matrix (ECM) impair wound healing are still elusive. Given the role of fibroblasts to produce a provisional matrix during tissue closure, we hypothesize that senescent human dermal fibroblasts produce a senescent-associated ECM that has a distinct composition and tissue architecture compared to non-senescent fibroblasts which delays wound closure.
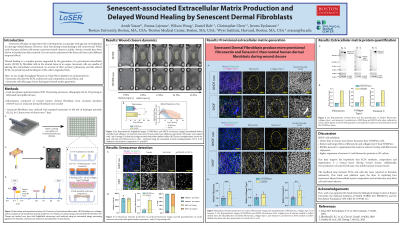
Methods: To address this hypothesis, we leveraged our 3D wound-on-chip microtissue platform to study the effect of senescent cells on stromal tissue repair and ECM architecture. Microfabricated devices consisting of 4 micropillars were cast in a 96-well plate using polydimethylsiloxane (PDMS). We seeded normal or senescent human dermal fibroblasts from neonatal foreskin in a rat-tail collagen type I matrix and allowed them to self-assemble into anchored microtissues. Senescence was induced in the normal fibroblasts through multiple exposures to hydrogen peroxide and confirmed with positive stains for p21, γ-H2AX, and senescent-associated-β-galactosidase. Microtissues were then ablated to create full-thickness wounds using a pulsed nanosecond laser. The provisional ECM was visualized with immunofluorescence labeling and confocal microscopy.
Results: We found that microtissues composed of senescent human dermal fibroblasts exhibited significantly delayed wound closure (41 ± 6.27 hours, n=9, N=3) when compared to normal human dermal fibroblasts from neonatal foreskin (26.2 ± 3.53 hours, n=9, N=3). Senescent fibroblasts deposited more fibronectin, collagen type I, collagen type III, and tenascin-C in the provisional matrix than normal fibroblasts. Interestingly, confocal microscopy also revealed that senescent microtissues contained larger, aligned fiber bundles of all four ECM components compared to the fine, mesh-like deposition in normal fibroblast microtissues. Notably, tenascin-C was deposited in small globules in normal fibroblast microtissues, highlighting the stark organizational differences between these fibroblast phenotypes.
Conclusion: Together, these data demonstrate that senescent cells produce a senescent-associated matrix with a tissue architecture that is distinct from the ECM produced by non-senescent fibroblasts. This may contribute to the delayed healing observed in senescent microtissues. Our 3D microtissue model provides a robust platform to further study the role of stromal cell ECM deposition during tissue repair, with control over cell populations and ECM density and composition.
Methods: To address this hypothesis, we leveraged our 3D wound-on-chip microtissue platform to study the effect of senescent cells on stromal tissue repair and ECM architecture. Microfabricated devices consisting of 4 micropillars were cast in a 96-well plate using polydimethylsiloxane (PDMS). We seeded normal or senescent human dermal fibroblasts from neonatal foreskin in a rat-tail collagen type I matrix and allowed them to self-assemble into anchored microtissues. Senescence was induced in the normal fibroblasts through multiple exposures to hydrogen peroxide and confirmed with positive stains for p21, γ-H2AX, and senescent-associated-β-galactosidase. Microtissues were then ablated to create full-thickness wounds using a pulsed nanosecond laser. The provisional ECM was visualized with immunofluorescence labeling and confocal microscopy.
Results: We found that microtissues composed of senescent human dermal fibroblasts exhibited significantly delayed wound closure (41 ± 6.27 hours, n=9, N=3) when compared to normal human dermal fibroblasts from neonatal foreskin (26.2 ± 3.53 hours, n=9, N=3). Senescent fibroblasts deposited more fibronectin, collagen type I, collagen type III, and tenascin-C in the provisional matrix than normal fibroblasts. Interestingly, confocal microscopy also revealed that senescent microtissues contained larger, aligned fiber bundles of all four ECM components compared to the fine, mesh-like deposition in normal fibroblast microtissues. Notably, tenascin-C was deposited in small globules in normal fibroblast microtissues, highlighting the stark organizational differences between these fibroblast phenotypes.
Conclusion: Together, these data demonstrate that senescent cells produce a senescent-associated matrix with a tissue architecture that is distinct from the ECM produced by non-senescent fibroblasts. This may contribute to the delayed healing observed in senescent microtissues. Our 3D microtissue model provides a robust platform to further study the role of stromal cell ECM deposition during tissue repair, with control over cell populations and ECM density and composition.

.jpeg)
