WHS Posters
(WHS-P23) Dermal Fibroblasts Contribute to Oxidative Stress in Diabetes
Friday, May 17, 2024
7:30 AM - 5:00 PM East Coast USA Time
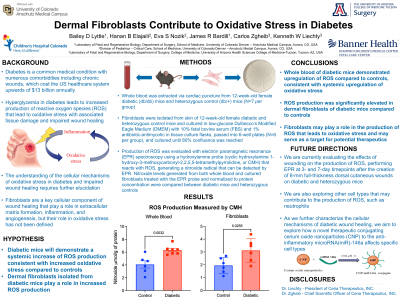
Background Diabetes is a common medical condition with numerous comorbidities including chronic wounds. Impaired wound healing in diabetes has been associated with inflammation and oxidative stress secondary to reactive oxygen species (ROS), which have traditionally been measured by evaluating dysregulation in enzymes that produce ROS. ROS also act as second messengers for cell populations present within diabetic wounds to stimulate further inflammation, resulting in a cycle of chronic inflammation and oxidative stress. Fibroblasts are a key cellular population involved in wound healing and have historically been identified to play a role in extracellular matrix formation, inflammation, and angiogenesis. Given that ROS interact with other cell populations beyond inflammatory cells, we theorized that fibroblasts may interact with ROS beyond their known cellular function and contribute to the overall milieu of the wound. We therefore hypothesized that both whole blood and dermal fibroblasts isolated from diabetic mice would demonstrate increased ROS production as measured by electron paramagnetic spectroscopy (EPR), which allows for direct measurement of ROS rather than just enzymatic markers. Methods Fibroblasts were isolated from skin of 12-week-old female diabetic (db/db) and heterozygous (db/+) control mice (N=1 per group) and cultured in low-glucose Dulbecco’s Modified Eagle Medium (DMEM) with 10% fetal bovine serum (FBS) and 1% antibiotic-antimycotic in tissue culture flasks. Once confluence was achieved, fibroblasts were passed into 6-well plates (N=6 per group) and cultured until 80% confluence was reached. Whole blood was extracted from 12-week-old female diabetic and heterozygous control mice (N=2 per group). Production of ROS was evaluated with electron paramagnetic resonance (EPR) spectroscopy using a hydroxylamine probe that upon the reaction with ROS generates a nitroxide radical that can be detected by EPR. Nitroxide levels generated from both whole blood and cultured fibroblasts treated with the EPR probe and normalized to protein concentration were compared between diabetic mice and heterozygous controls. Results Fibroblasts from diabetic mice demonstrated significantly higher ROS production compared to heterozygous controls (p=0.042). Similarly, whole blood from diabetic mice demonstrated significantly higher ROS production compared to heterozygous controls (p=0.047). Conclusions ROS production was significantly elevated in dermal fibroblasts of diabetic mice compared to controls, suggesting that fibroblasts may play a role in the production of ROS that leads to oxidative stress. Additionally, whole blood of diabetic mice demonstrated upregulation of ROS, suggesting the presence of a systemic effect. Dermal fibroblasts remain a key regulator of impaired wound healing in diabetes due to multiple mechanisms and may serve as a target for potential therapeutics.

.jpeg)
