Laboratory Research
(LR-028) Dehydrated Human Amnion Chorion Membranes Contain Key Extracellular Matrix Proteins, Resist Rapid In Vitro Degradation, and Function as a Cell Scaffold
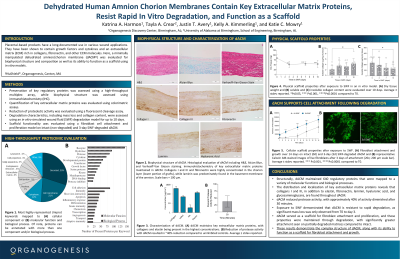
Methods:
dACM biophysical structure was evaluated with immunohistochemistry and protein composition using a high-throughput multiplex array. The ability of dACM to inhibit protease activity was evaluated using a fluorescent cleavage assay. dACM was subjected to in vitro degradation in a simulated wound fluid (SWF) model for up to 10 days to determine mass loss and collagen content. The impact of degradation on functionality was measured by an in vitro primary human dermal fibroblast cell attachment model. Intact (non-degraded) and 3-day SWF-degraded dACM were seeded, cultured for up to 10 days, and evaluated for proliferation.
Results:
Key ECM proteins including collagens I and III and fibronectin were highly concentrated in the chorion layer, while laminin was predominately found in the basement membrane of the amnion. Proteomic evaluation identified 640 regulatory proteins present in dACM that were mapped to a variety of molecular functions and biologic processes. dACM inhibited protease activity, with approximately 40% of activity diminished after 30 minutes. When exposed to SWF, there was a significant mass loss at day 3, but no substantial degradation from days 3-7 and days 7-10. Overall, degradation resulted in 37.6% loss and a significant increase in released soluble collagen throughout the study. Initial fibroblast attachment was seen on both intact and degraded dACM, with degraded samples exhibiting significantly more attachment. While cells proliferated on both, there was significantly greater proliferation on 3-day degraded dACM on days 1, 3, 7, and 10 compared to intact; this result is suspected to be due to the increased availability of binding sites revealed from degradation.
Discussion:
These results demonstrate the complex structure and composition of dACM, along with its ability to withstand rapid degradation and function as a scaffold for fibroblast attachment and growth throughout expected degradation processes.

.jpeg)