Clinical Research
(CR-026) Transforming Powder Dressing (TPD) for Diabetic Ulcer Care
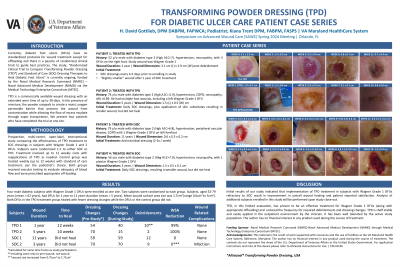
Currently, diabetic foot ulcers (DFU) have no standardized protocols for wound treatment except for offloading. The study, “Randomized Clinical Trial to Compare Transforming Powder Dressing (TPD) and Standard of Care (SOC) Dressing Therapies to Heal Diabetic Foot Ulcers” is currently ongoing, funded by the Naval Medical Research Command (NMRC) – Naval Advanced Medical Development (NAMD) via the Medical Technology Enterprise Consortium (MTEC). TPD is a commercially available, FDA registered dressing for wounds that can be left in place for up to 30 days. In the presence of moisture, the powder creates a semipermeable barrier which promotes wound healing as well as preventing contamination of the open tissues. We present 4 patients who have completed the trial at one site.
Methods: Prospective, multi-center, open-label, interventional study comparing the effectiveness of TPD treatment to SOC dressings in subjects with Wagner Grade 1 and 2 DFUs. Subjects were randomized 1:1 to either SOC or TPD. Treatment involved weekly visits with reapplications of TPD as needed. Control group was treated weekly with standard of care treatment, per the podiatrist’s choice. Both groups received appropriate off-loading.
Results: Four male diabetic subjects with Wagner Grade 1 DFUs were enrolled at one site. Two subjects were randomized to each group. Subjects, aged 52-79 years, had DFUs for 3 months to 11 years duration. Mean wound size was 1.7 cm2. All DFUs in the TPD treatment group healed. DFUs in the control group did not.
Discussion:
The initial observations of our patient population involved in this study indicate that implementation of TPD treatment in subjects with Wagner Grade 1 DFUs refractory to SOC result in overall wound healing and subject satisfaction. Analysis of additional subjects enrolled in this study will be performed upon study close-out.
TPD, in this limited evaluation, has proven to be an effective treatment for Wagner Grade 1 DFUs along with appropriate offloading. TPD is shelf stable and easily applied in the outpatient environment by the clinician. It is well tolerated by the active study population. The author has no financial interest in any product used during the course of treatment.

.jpeg)