Laboratory Research
(LR-027) Investigating the Rapid Antimicrobial Action of a Silicone PHMB Foam Dressing
With a multitude of dressings in the wound care market, it is important for a healthcare provider to administer the most appropriate product. The ability for rapid and sustained antimicrobial activity of an antimicrobial dressing is a significant benefit to wound management and is a vital component to consider.
A Silicone PHMB foam dressing has been developed, that is indicated for moderate to heavily exuding chronic and acute wounds that are infected or at risk of infection, and is designed to provide secure, non-irritating adhesion, whilst supporting non-traumatic removal. The PHMB contained within the dressing is a broad spectrum antimicrobial substance, whereby it selectively binds and condenses chromosomes of the bacteria, resulting in cell death.
The following study aims to demonstrate the rapid antimicrobial performance of the device, and another PHMB containing foam product, against a range of common wound isolates.
Methods:
The antimicrobial performance was assessed via a direct inoculation assay in accordance with AATCC TM100 guidelines, over a 1hr to 7 day period. Conditions were applied to replicate clinical wound conditions and the challenge organisms utilised represent microbial diversity, as found within chronic wounds.
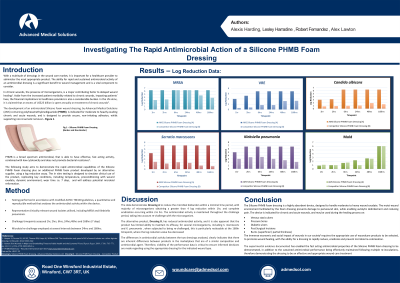
Results:
The data demonstrates the device to have excellent antimicrobial activity, with complete eradication being obtained within 1hr to 2hrs for the majority of microorganisms tested, with minimal performance being observed for the second device tested.
Discussion:
Antimicrobial activity can be highly variable depending on the dressing composition and antimicrobial agent. The in vitro testing has enabled the fast acting antimicrobial properties of the Silicone PHMB foam dressing to be demonstrated, therefore highlighting the product to be an effective and appropriate wound care treatment.

.jpeg)