Laboratory Research
(LR-044) Lyophilized Human Amnion and Chorion Membrane Modulates the Macrophage Inflammatory Response
Methods:
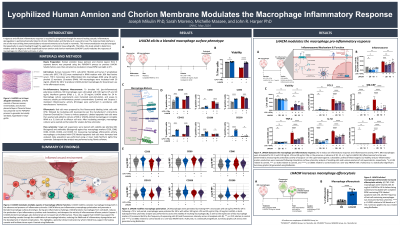
LHACM allografts from 5 donated placentae were prepared using the PURION process, consisting of gentle cleansing, followed by lyophilization and terminal sterilization. Human THP-1 macrophages were challenged with lipopolysaccharide (LPS) and human interferon gamma (IFNγ), in the presence or absence of various doses of LHACM eluate, and subsequently harvested for measurement of viability by flow cytometry. Pro-inflammatory protein production was measured via Luminex analysis of supernatants and inflammasome activation was measured by addition of a Caspase-1 cleavage-activated Luciferase reporter. Additionally, macrophages grown in the presence or absence of LHACM were fed CFSE-labeled dead Jurkat cell debris to measure efferocytosis.
Results:
LHACM decreases macrophage death amid pro-inflammatory stimulation with LPS and IFNγ and exhibits no cytotoxic effect on resting macrophages at the concentrations tested. LHACM-treated macrophages demonstrate a reduction in pro-inflammatory proteins such as TNFα, IL-1β, and IL-6 produced in response to inflammatory stimulus. In a dose-dependent manner, LHACM reduces the Caspase-1 activity downstream of the inflammasome activation associated with the pro-inflammatory response. Finally, LHACM increases the rate at which macrophages consume dead cell debris.
Discussion:
These data demonstrate that LHACM modulates significant features of inflammatory macrophage biology while enhancing pro-reparative functions including, efferocytosis and cell survival. This suggests that the LHACM may support the wound healing cascade through the modification of macrophage behavior that lead to a reduction in the likelihood of inflammatory dysregulation and the development of related complications. This highlights a potential clinical mechanism by which LHACM may support the healing cascade and facilitate tissue repair.

.jpeg)
